|
Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. For instance they discovered phosphorus when they isolated it from urine.  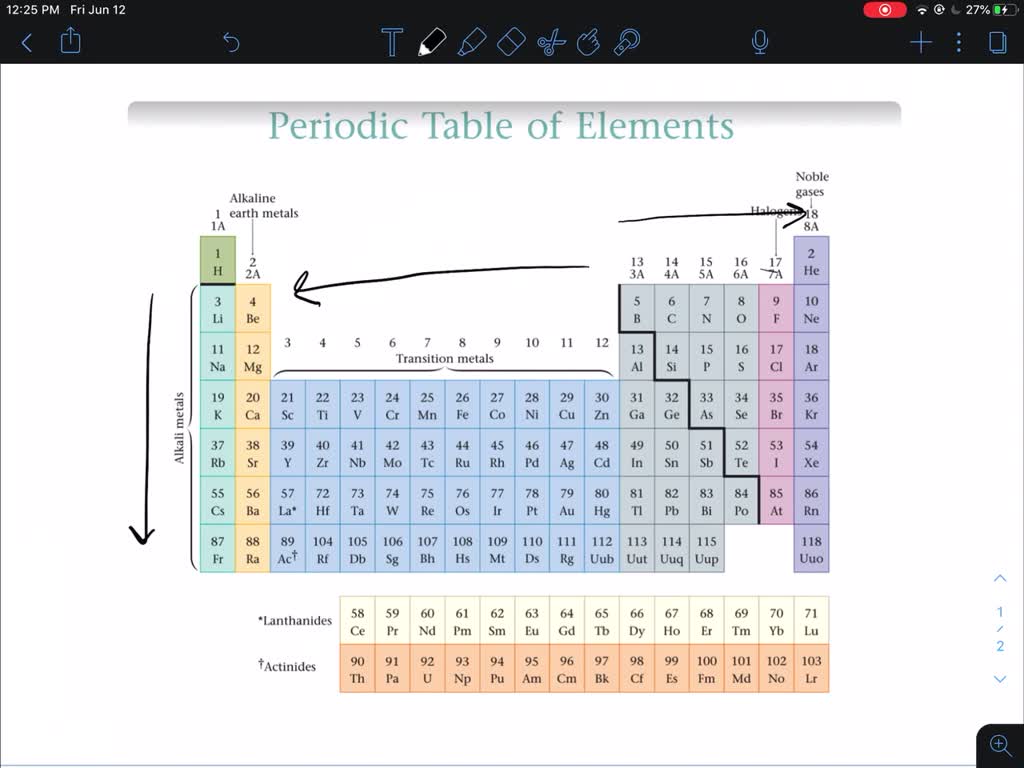
Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. 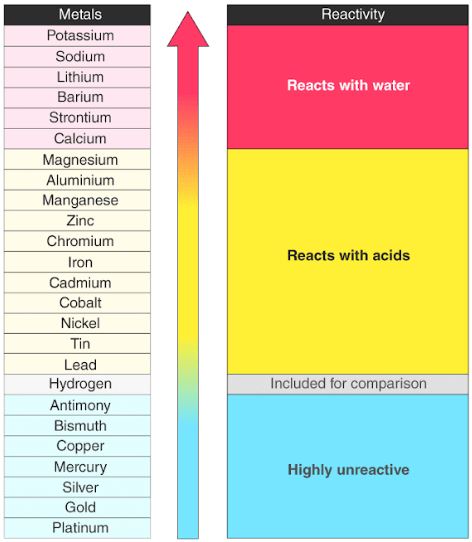
He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Transition Metals: Groups 3-12 - d and f block metals have 2 valence electrons. Alkaline Earth Metals: Group 2 (IIA) - 2 valence electrons. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. Another common method of categorization recognizes nine element families: Alkali Metals: Group 1 (IA) - 1 valence electron. Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. 2: Elements categorized into metals, non-metals and metalloids. The oxidation number of an element in this group can range from +5 to -2, depending on the group in which it is located. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. The physical properties of metalloids tend to be metallic, but their chemical properties tend to be non-metallic. This is primarily a function of the smaller size of the first member of each group, which allows better overlap of atomic orbitals.Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years. Remember that an element exhibits a positive oxidation state when combined with a more electronegative element and that it exhibits a negative oxidation state when combined with a less electronegative element.Īnother difference between the first group member and subsequent members is the greater ability of the first member to form π bonds. The greatest boiling and melting details are associated with cross over metals. These reactive components usually form ores along with other metals and non-precious metals. For example, nitrogen forms only NF 3, whereas phosphorus forms both PF 3 and PF 5. In the Periodic Kitchen table, changeover metals are normally numerous elements in the earth’s crust. 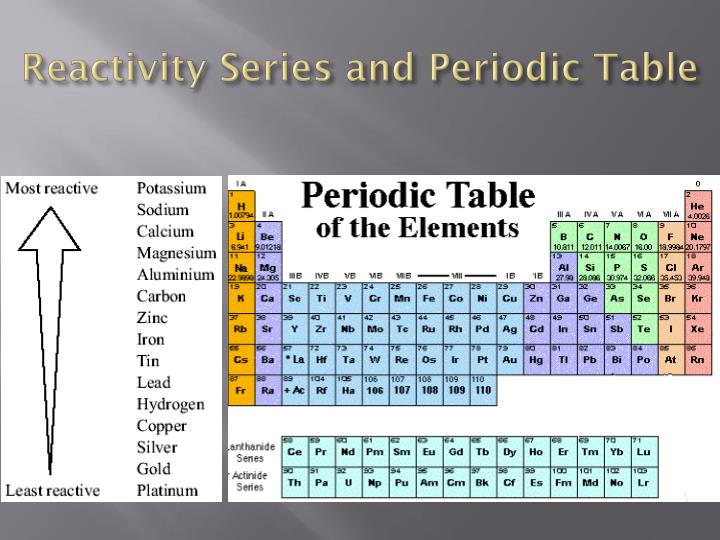
The reasons for this include smaller size, greater ionization energy, and (most important) the fact that the first member of each group has only four valence orbitals (one 2 s and three 2 p) available for bonding, whereas other group members have empty d orbitals in their valence shells, making possible five, six, or even more bonds around the central atom. The first member of each nonmetal group exhibits different behaviors, in many respects, from the other group members. Sodium (Na) and potassium (K) are some of the most reactive metals. Reactivity of Elements of the Periodic Table Novice 1 m Reactivity is a measure of how easily an element will combine with other elements to form compounds. Reactivity: Metals are very reactive, some more than others, but most form compounds with other elements quite easily. Silver (Ag) and copper (Cu) are some of the most efficient metals and are often used in electronics. The activity series is a list of elements in decreasing order of their reactivity. Because of the alkali metals electron configuration (wants to give its one electron to another substance), theyre highly reactive. Therefore, it is useful to have a list of elements in order of their relative reactivity. \): Nonmetals exhibit these common oxidation states in ionic and covalent compounds. Conduction: Metals are good at conducting electricity. The most reactive metals in the periodic tables are the alkali metals, followed by the alkaline earth metals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
